Associate Professor UCHIYAMA Masahiko
Faculty, Affiliation
Research Center for Experimental Modeling of Human Disease
College and School Educational Field
Division of Frontier Engineering, Graduate School of Natural Science and Technology
Division of Natural System, Graduate School of Natural Science and Technology
School of Pharmaceutical Sciences, College of Medical, Pharmaceutical and Health Sciences
Laboratory
Research Institute for Instrumental Analysis TEL:076-234-4428 FAX:076-234-4428
Academic Background
【Academic background(Doctoral/Master's Degree)】
Kanazawa University Doctor Graduate School of Natural Science & Technology 199503 Unfinished course
【Academic background(Bachelor's Degree)】
Kanazawa University 199103
Career
Tokyo University of Pharmacy and Life Science School of Pharmacy(1995/04/01-2002/03/31)
Kanazawa University Faculty of Pharmaceutical Sciences(2002/04/01-)
Kanazawa university Advanced Science Research Center(2006/05/01-)
Year & Month of Birth
1967/04
Academic Society
The Pharmaceutical Society of Japan
The Society of Synthetic Organic Chemistry, Japan
Award
Specialities
Development of environmentally benign synthetic reactions、Chemical pharmacy、Organic chemistry、Synthetic organic chemistry
Speciality Keywords
Green Chemistry, Environment, Resources, Synthetic Organic Chemistry
Research Themes
Synergistic Cu(I)/Ga(III) catalyzed Conia-ene reaction
Conia-ene Reaction is originally known as the thermal intramolecular ene reaction of enolizable carbonyl compounds with tethered alkene or alkyne moiety. The utility of this reaction has been limited by its harsh reaction conditions. Recently much attention has been directed to the Conia-ene reaction using various metal catalysts, which has allowed the reaction to proceed under mild conditions. We investigated the catalytic Conia-ene reaction with a focus on cationic Cu(I) as alkyne activator. As a result, synergistic Cu(I)/Ga(III) catalytic system for the Conia-ene reaction has been developed. In the presence of CuOTf・1/2C6H6 and Ga(OTf)3, various acetylenic 1,3-dicarbonyl compounds underwent the Conia-ene reaction under mild conditions in moderate to good yields.
Study on Asymmetric Reactions using Optically Active Organoselenium Reagents
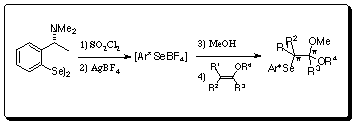 Oxyselenenylation reactions involving olefin with the trans-addition of electrophilic selenium reactant and oxygen nucleophile, have been used frequently as they can stereoselectively introduce two kinds of functional group onto the two carbon atoms of olefin at once. In addition, since the reaction develops into an asymmetric reaction through the use of an optically active selenium reactant, its utility is improving. However, because the substrate of the asymmetric reaction has been limited to simple olefin, we used vinyl ether as a substrate and found that optically active acetal can be synthesized. We subsequently conducted synthesis of a variety of natural products with a new synthesis method that involves using the optically active acetal that is obtained.
Oxyselenenylation reactions involving olefin with the trans-addition of electrophilic selenium reactant and oxygen nucleophile, have been used frequently as they can stereoselectively introduce two kinds of functional group onto the two carbon atoms of olefin at once. In addition, since the reaction develops into an asymmetric reaction through the use of an optically active selenium reactant, its utility is improving. However, because the substrate of the asymmetric reaction has been limited to simple olefin, we used vinyl ether as a substrate and found that optically active acetal can be synthesized. We subsequently conducted synthesis of a variety of natural products with a new synthesis method that involves using the optically active acetal that is obtained.
Study on Ce(IV)-catalyzed asymmetric oxidative radical reaction
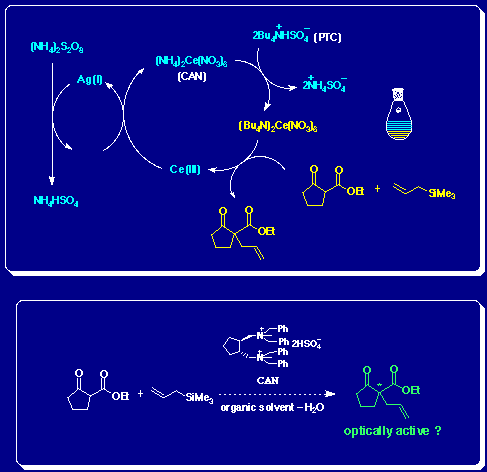 Oxidative radical C-C bond forming reaction with ceric ammonium nitrate (CAN) has only been induced in limited polar solvents, such as water, methanol, and acetonitrile, due to CAN’s solubility. We investigate the hypothesis that the reaction could be induced in the organic phase if quaternary ammonium salt is added as a phase transfer catalyst (PTC) in a 2-phase system consisting of water and nonpolar solvent.In addition, we conduct research on canalization of the Ce(IV) reactant used and desymmetrization with optically-active PTC.
Oxidative radical C-C bond forming reaction with ceric ammonium nitrate (CAN) has only been induced in limited polar solvents, such as water, methanol, and acetonitrile, due to CAN’s solubility. We investigate the hypothesis that the reaction could be induced in the organic phase if quaternary ammonium salt is added as a phase transfer catalyst (PTC) in a 2-phase system consisting of water and nonpolar solvent.In addition, we conduct research on canalization of the Ce(IV) reactant used and desymmetrization with optically-active PTC.
Study on Asymmetric Reduction of Symmetric Diketones with Baker's Yeast
Study on Oxidative Radical Cyclization using Polymer-supported Mn(III) reagent
Books
Papers
- A simple enantioselective synthesis of (R)- and (S)-1,7-dioxaspiro[5.5]undecane via intramolecular asymmetric oxyselenenylation TETRAHEDRON LETTERS 42 10 1931 2001/03
- Asymmetric methoxyselenenylation of alkyl vinyl ethers: a new route to chiral acetals TETRAHEDRON LETTERS 42 8 1559 2001/02
- Diastereocomplementary construction of optically active bicyclo[4.3.0]nonenone skelton based on Pauson-Khand reaction TETRAHEDRON LETTERS 39 43 7909 1998/10
- Highly enantioselective reduction of symmetrical diacetylaromatics with baker\'s yeast Tetrahedron: Asymmetry 8 20 3467 1997/10
- Stereoselective construction of optically active bicyclo[3.3.0]octenone derivatives based on the Pauson-Khand reaction JOURNAL OF THE CHEMICAL SOCIETY-PERKIN TRANSACTIONS 1 17 2903 1998/09
- Stereoselective synthesis of optically active perhydrofuro[2,3-b]furan derivatives TETRAHEDRON LETTERS 42 28 4653 2001/07
- Stereoselective total syntheses of (┣D3+(/)-┫D3)-arthrinone and related natural compounds TETRAHEDRON LETTERS 41 51 10013 2000/12
- A Highly Diastereoselective Construction of Optically Active Bicyclo[3.3.0]octenone Derivatives from L-Ascorbic Acid via Pauson-Khand Reaction TETRAHEDRON LETTERS 36 32 5761 1995/06
- Bis(cyclopentadienyl)tetracarbonyldimolybdenum-alkyne Complexes Mediated [2+2+1]Cycloaddition: the Formation of 3-Substituded Cyclopentenone Derivatives CHEMICAL COMMUNICATIONS 14 1014 1992/04
- Concise Synthesis of the Tricyclic Skeleton of Cylindricines Using a Radical Cascade Involving 6-Endo Selective Cyclization SYNLETT 7 1179-1181 2005/05
- 7-Endo Selective Aryl Radical Cyclization onto Enamides Leading to 3-Benzazepines: Concise Construction of a Cephalotaxine Skeleton JOURNAL OF ORGANIC CHEMISTRY 70 5 1922-1925 2005/03
- A short synthesis of lennoxamine using a radical cascade ORGANIC LETTERS 7 20 4389-4390 2005/09
- Radical cyclization in 1,4-dimethylpiperazine TETRAHEDRON LETTERS 47 35 6263-6266 2006/08
- Stereoselective synthesis of trans-3a-aryloctahydroindoles using cyclization of N-vinylic α-(methylthio)acetamides. HETEROCYCLES 69 1 69-72 2006
Conference Presentations
Others
Arts and Fieldwork
Patent
Theme to the desired joint research
Grant-in-Aid for Scientific Research
○「固相担持型Mn(III)反応剤を用いる酸化的ラジカル環化反応の開発とその応用」(2003-2004)
○「Ce(IV)とフッ素アニオンを用いる有機ケイ素化合物からの新規ラジカル発生法の開発」(2008-2010)
Competitive research funding,Contribution
Collaborative research,Consignment study
Classes (Bachelors)
○Graduation Research in Pharmacy 1(2023)
○Seminar in Pharmacy 1(2023)
○Laboratory Rotation 3(2023)
○Basic Medical and Pharmaceutical Sciences Rotation Practice(Course in Pharmaceutical Sciences)Ⅲ(2023)
○Experiments in Organic Chemistry(2023)
○Spectroscopic Methods in Organic Chemistry(2023)
○Spectroscopic Methods in Organic Chemistry(2023)
○Seminar in Organic Chemistry 4(2023)
○Applied Organic Chemistry 2(2023)
○Organic Chemistry 4(2023)
○Seminar in Applied Organic Chemistry 2(2023)
○Organic Chemistry 4(2022)
○Applied Organic Chemistry 2(2022)
○Seminar in Organic Chemistry 4(2022)
○Seminar in Applied Organic Chemistry 2(2022)
○Spectroscopic Methods in Organic Chemistry(2022)
○Experiments in Organic Chemistry(2022)
○Basic Medical and Pharmaceutical Sciences Rotation Practice(Course in Pharmaceutical Sciences)Ⅲ(2022)
○Experiments in Organic Chemistry(2021)
○Spectroscopic Methods in Organic Chemistry(2021)
○Seminar in Organic Chemistry 4(2021)
○Organic Chemistry 4(2021)
○Organic Chemistry 4(2020)
○Spectroscopic Methods in Organic Chemistry(2020)
○Seminar in Organic Chemistry 4(2020)
○Experiments in Organic Chemistry(2020)
○Graduation Research in Pharmaceutical Sciences 1(2020)
○Seminar in Pharmaceutical Sciences(2020)
○Graduation Research in Pharmaceutical Sciences 1(2019)
○Seminar in Pharmaceutical Sciences(2019)
○Graduation Research in Pharmaceutical Sciences 2(2019)
○Experiments in Organic Chemistry(2019)
○Seminar in Organic Chemistry 4(2019)
○Organic Chemistry 4(2019)
○Spectroscopic Methods in Organic Chemistry(2019)
○Graduation Research in Pharmaceutical Sciences 1(2018)
○Laboratory Rotation 3(2018)
○Seminar in Pharmaceutical Sciences(2018)
○Experiments in Organic Chemistry(2018)
○Seminar in Organic Chemistry 4(2018)
○Spectroscopic Methods in Organic Chemistry(2018)
○Organic Chemistry 4(2018)
○Experiments in Organic Chemistry(2017)
○Organic Chemistry 4(2017)
○Seminar in Organic Chemistry 4(2017)
○Spectroscopic Methods in Organic Chemistry(2017)
○Spectroscopic Methods in Organic Chemistry(2016)
○Organic Chemistry 4(2016)
○Seminar in Organic Chemistry 4(2016)
○Experiments in Organic Chemistry(2016)
Classes (Graduate Schools)
○Learning Recent Advancement in Pharmaceutical Sciences(2023)
○Research Training in Pharmaceutical Sciences(2023)
○Pharmaceutical Sciences Practice II(2023)
○Interdisciplinary Seminar(2023)
○Experimental Techniques in Pharmachemistry(2023)
○Green Organic Chemistry(2022)
○Learning Recent Advancement in Pharmaceutical Sciences(2022)
○Research Training in Pharmaceutical Sciences(2022)
○Pharmaceutical Research Practice I(2022)
○Pharmaceutical Sciences Practice II(2022)
○Interdisciplinary Seminar(2022)
○Pharmaceutical Sciences Practice I(2022)
○Advanced Seminar on Pharmachemistry(2022)
○Experimental Techniques in Pharmachemistry(2022)
○Advanced Seminar on Pharmachemistry(2021)
○Pharmaceutical Sciences Practice II(2021)
○Pharmaceutical Research Practice II(2021)
○Research Training in Pharmaceutical Sciences(2021)
○Learning Recent Advancement in Pharmaceutical Sciences(2021)
○Pharmaceutical Research Practice I(2021)
○Pharmaceutical Sciences Practice I(2021)
○Experimental Techniques in Pharmachemistry(2021)
○Interdisciplinary Seminar(2020)
○Pharmaceutical Sciences Practice I(2020)
○Pharmaceutical Research Practice I(2020)
○Advanced Seminar on Pharmachemistry(2020)
○Experimental Techniques in Pharmachemistry(2020)
○Pharmaceutical Sciences Practice II(2019)
○Research Training in Pharmaceutical Sciences(2019)
○Learning Recent Advancement in Pharmaceutical Sciences(2019)
○Pharmaceutical Research Practice II(2019)
○Experimental Techniques in Pharmachemistry(2019)
○Green Organic Chemistry(2018)
○Pharmaceutical Research Practice I(2018)
○Pharmaceutical Sciences Practice I(2018)
○Advanced Seminar on Pharmachemistry(2018)
○Experimental Techniques in Pharmachemistry(2018)
○Research Training in Pharmaceutical Sciences(2018)
○Learning Recent Advancement in Pharmaceutical Sciences(2018)
○Experimental Techniques in Pharmachemistry(2017)
○Advanced Organic Chemistry V(2016)
○Green Organic Chemistry(2016)



