Professor MATSUSHITA Ryo
Faculty, Affiliation
Faculty of Pharmacy, Institute of Medical, Pharmaceutical and Health Sciences Faculty of Pharmacy, Institue of Medical, Pharmaceutical and Health Sciences
College and School Educational Field
Division of Electrical,Information and Communication Engineering, Graduate School of Natural Science and Technology
Division of Cancer Medicine, Graduate School of Medical Science
Division of Natural System, Graduate School of Natural Science and Technology
School of Pharmacy, College of Medical, Pharmaceutical and Health Sciences
School of Pharmaceutical Sciences, College of Medical, Pharmaceutical and Health Sciences
Laboratory
Clinical Drug Informatics TEL:076-234-4405 FAX:076-234-4405
Academic Background
【Academic background(Bachelor's Degree)】
Kanazawa University 198203
【Degree】
PhD
Career
(1982/04/01-1983/03/31)
Kanazawa University Hospital Pharmacy(1997/04/01-2000/03/31)
Kanazawa University Natural Science and Technology(2000/04/01-2009/03/31)
Kanazawa University Natural Science and Technology(2009/04/01-)
Kanazawa University Faculty of Pharmacy(2010/04/01-)
Year & Month of Birth
1959/12
Academic Society
Japanese Society for the Study of Xenobiotics
The Japanese Society of Therapeutic Drug Monitoring
Award
Specialities
Medical pharmacy、Biological pharmacy、Clinical Pharmacokinetics、Medical pharmacy、Epidemiology and preventive medicine
Speciality Keywords
Clinical Pharmacokinetics Clinical Pharmacology Clinical Pharmacy Therapeutic Drug Monitoring Pharmaceutics Pharmacuticals
Research Themes
Clinical pharmaceutical or phamracokinetic study for drug therapeutics and adverse event monittoring
Therapeutic Drug Monitoring (TDM) can provide tailor-made therapy. We aim to construct the TDM systems with pharmacokinetics, population pharmacokinetics, disease animal models and molecular biology for the contribution of rational drug therapy.
Assistance for pharmacist performing pharmaceuitcal care by scientific approach
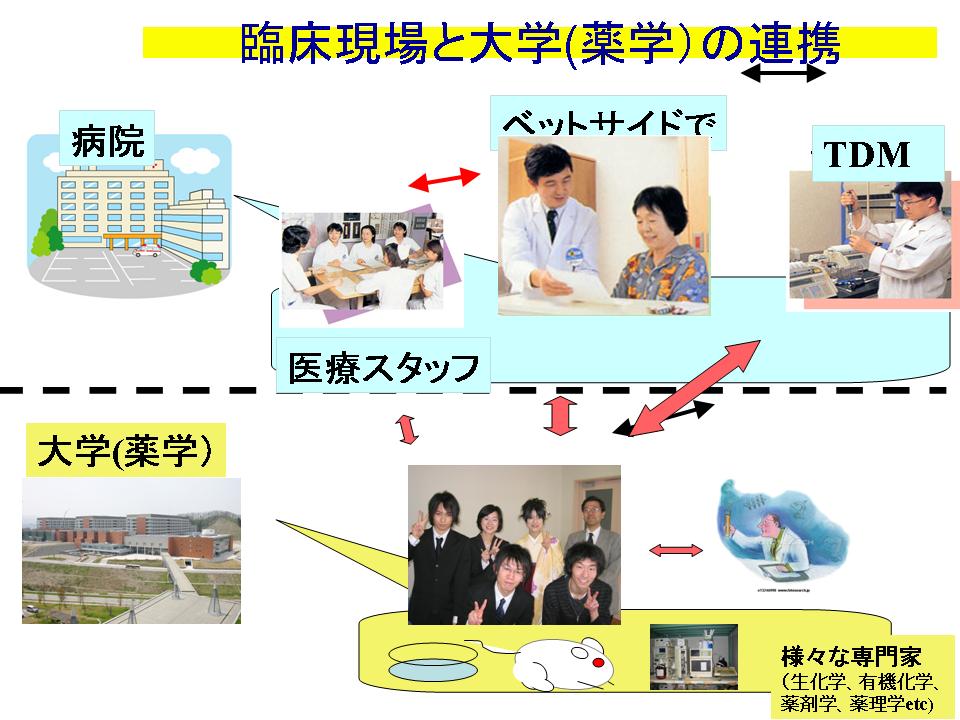 Variou clinical problems have plagued pharmacists in clinical fields. Then, I hope to assist for them by establishing the clincal evidences with scientific approach.
Variou clinical problems have plagued pharmacists in clinical fields. Then, I hope to assist for them by establishing the clincal evidences with scientific approach.
Pharmacokinetic Analyses of Interndividual Differences in Diseased states
Study and application of therapeutic drug monitoring
Books
- Yukio Kato New Dosage Form: Drug Delivery System 359-382 2007/04
- Hiroaki Okada Liquid Pharmaceuticals, Sterile Pharmaceuticals 218-243 2007/09
- Hitoshi Sato PK-PD review 209-222 2008/01
Papers
- Effect of meropenem on disposition kinetics of valproate and its metabolites in rabbits Pharm Res 18 9 1320-1326 2001/09
- Kinetic phenotypic diagnosis of N-acetylation polymorphism in patients based on ratio of urinary metabolites of salicylazosulfapyridine Int J Pharm 229 1-2 183-191 2001/01
- Synergistic antitumor interaction of human monocyte chemotactant protein-1 gene transfer and modulator for tumor-infiltrating macrophages Pharm Res 15 5 685-689 1998/05
- Synergistic effect of indomethacin and bleomycin on tumor growth produced by activating antitumor immunity Pharm Res 18 2 243-245 2001/02
- Synergistic effect of indomethacin with adriamycin and cisplatin on tumor grouwth Bio Pharm Bull 24 10 1214-1217 2001/10
- A candidate for cancer gene therapy: the effects of MIP-1α gene transfer to an adenocarcinoma cell line on tumorigeneicity and protecitive immunity in mice The symposium on the impact of biotechnology on cancer prognosis, diagnosis and prevention 1998/10
- Concomitant cancer chemotherapy with gene pharmaceuticals Controlled relase society(CRS)-Chiniese pharmaceutical association (CPA) joint workshop on recent advances in drug delivery science and technology 1997/09
- Kinetic analysis of the Interaction between valproic acid and meropenem in rabbits International Conference on Drug Interaction 73 2000/09
- Synergistic effect of anticancer drug and indomethacin to Colon 26 clone 20 tumor-bearing mice XIIIth international congress of pharmacology 1998/07
- The Challenge of implementing the Educational Problems Related to Drug Therapy in Master’s Course at Kanazawa University Hospital in Japan Pharmacy World Congress’99, 59th International Congress of FIP 1999/09
- The Clinical Pharmacy Education Curriculum Development for the Students of the Graduate School of Kanazawa University, The 3rd East Asia Conference on Developing Clinical Pharmacy Practice and Education 32 2001/07
- Causative agent of vascular pain among photodegradation products of dacarbazine JOURNAL OF PHARMACY AND PHARMACOLOGY 54 1117-1122 2002/08
- Evaluation of predictablility for vancomycin dosage regimens by the Bayesian method with Japanese population pharmacokinetic parameters BIOLOGICAL & PHARMACEUTICAL BULLETIN 25 10 1333-1338 2002/10
- Surveillance of antibiotic usage to make a new gude for parmacists to the infection control in Kouseiren Chu-nou Hospital 28 4 339-346 2002/08
- A comparative study of the usefulness of Toki-shakuyaku-san and an oral iron preparation in the treatment of hypochromic anemia in cases of uterine myoma YAKUGAKU ZASSHI-JOURNAL OF THE PHARMACEUTICAL SOCIETY OF JAPAN 123 9 817-824 2003/09
- A comparative study of laboratory parameters and symptoms effected by Toki-shakuyaku-san and an iron preparation in rats iwth iron-deficiency anemia BIOLOGICAL & PHARMACEUTICAL BULLETIN 27 6 871-878 2004/06
- In vitro detection of mar1 mRNA in murine leukemia cells with 111In-labeled oligonucleotide 31 1523-1529 2004/11
- Influence of malignancy on the pharmacokinetics of vancomycin hydrochloride in Japanese MRSA patients after dosage adjustment with the Bayesian method Hitomi Tera,achi, Ryo Matsushita , Akira Tsuji 53 6 357-363 2005/06
- Verification at blood sampling time and administering time to obtain the best blood level index of NeoralTM. Naho Yamamoto, Ryo Matsushita, Koichi Yokogawa, Ken-ichi Miyamoto 25 2 69-74 2008/04
- The Usefulness of Inspiratory Flow Rate during Inhalation Corticosteroid Therapy in Asthma. Masashi Banno, Hidenori Ibata, Takashi Niimi, Shigeki Sato, Ryo Matsushita in press 2009/05
- Byakkokaninjinto prevents body water loss by increasing the expression of kidney aquaporin-2 and skin aquaporin-3 in KKAy mice T. Aburada, N. Ikarashi, M. Kagami, Y. Ichikawa, M. Sugitani, A.Maniwa, H. Ueda, T. Toda, K. Ito, W. Ochiai, R. Matsushita, K. Miyamoto, K. Sugiyama. Phytother Res 25 6 897-903 2011/06
- Study on prescription status and clinical utility of injectable antimicrobial agents by retrospective analysis of multicenter medical cost accouting data 32 6 851-862 2011/06
- Enhanced renal clearance of vancomycin in rats with carcinogen-induced osteosarcoma 1) I. Shimada , C. Iwata, S. Taga, H. Teramachi, M. Nomura, K. Miyamoto, H. Tsuciya, T. Wada, K. Kimura, R ANTICANCER RESEARCH 32 3 823-829 2012/03
- Pharmacist-based Donepezil Outpatient Consultation Service to improve medication persistence N. Watanabe, K. Yamamura, Y. Suzuki, H. Umegaki, K. Shigeno, R. Matsushita R, Y. Sai, Patient Prefer Adherence 6 605-611 2012/04
- Influence of long-term enteral nutrition on pharmacokinetics of digoxin in rats. Higashi K, Tanaka C, Imanishi K, Sawamoto K, Horikawa T, Ohkawa H, Matsushita R, Sai Y, Miyamoto K. Drug Metab Pharmacokinet. 28 1 44-52 2013/01
- Structural changes in albumin are a possible mechanism for fluctuation of cefazorin and ibuprofen plasma protein binding in rats with carcinogen-induced osteosarcoma. Itakura Y, Taga S, Iwata C, Teramachi H, Miyamoto K, Tsuchiya H, Wada T,Matsushita R. Anticancer Res. 35 4 2063-9 2014/04
- Pharmacokinetics and safety of 3,4-diaminopyridine base in healthy Japanese volunteers. Ishida N, Kobayashi E, Kondo Y, Matsushita R, Komai K. Int J Clin Pharmacol Ther 53 8 674-80 2015/08
Conference Presentations
Others
- Use of generic medicine promoting projects 2011/03
Arts and Fieldwork
Patent
Theme to the desired joint research
○Development of clinical pharmacy education with IT
○Study of therapeutic drug monitoring
Grant-in-Aid for Scientific Research
○「希少疾病LEMS治療薬3,4-ジアミノピリジン適正使用のための薬物動態学的検討」(2014-2016)
○「がん病態下における薬物の腎クリアランス亢進メカニズムの解明」(2011-2013)
○「インターネットを活用した医療薬学系大学院教育カリキュラムの開発」(2003-)
○「服薬指導データベース」(1998-)
○「服薬指導データベース」(1999-)
Competitive research funding,Contribution
Collaborative research,Consignment study
○Clinical pharmacy education development for the working students with internet(2000-2002)
Classes (Bachelors)
○Manufacturing Pharmacy(2017)
○Communication Skills in Clinical Pharmacology(2017)
○Community Pharmacy(2017)
○Medical Statistics(2017)
○Drug and Medical Informatics(2017)
○Pharmacoepidemiology(2017)
○Presentation and Debate (Freshman Seminar II)(2017)
○Seminar in Drug Therapeutics(2017)
○Right to Health and Medical Services Law(2017)
○Clinical Chemistry(2017)
○Community Pharmacy Practice 1(2017)
○Seminar in Clinical Pharmacy 2(2017)
○Basic Drug Development(2017)
○Seminar in Pharmaceutical Practice(2017)
○Hospital Pharmacy Practice 2(2017)
○Introduction to Active Listening(2017)
○Hospital Pharmacy Practice 1(2017)
○Community Pharmacy Practice 2(2017)
○Exercise of Therapeutic Drug Monitoring(2017)
○Introduction to Nursing(2017)
○Clinical Nutrition Support(2017)
○Introduction to Region-studies(2016)
○Management of Community Pharmacy(2016)
○General Pharmacy 2(2016)
○Seminar in Drug Therapeutics(2016)
○Community Pharmacy Practice 1(2016)
○Basic Drug Development(2016)
○Drug and Medical Informatics(2016)
○Hospital Pharmacy Practice 2(2016)
○Seminar in Clinical Pharmacy 2(2016)
○Manufacturing Pharmacy(2016)
○Seminar in Clinical Pharmacy 1(2016)
○Hospital Pharmacy Practice 1(2016)
○Community Pharmacy Practice 2(2016)
Classes (Graduate Schools)
○Risk Management Practice for Cancer Patients(2017)
○Seminar in Monitoring the Adverse Events of Cancer Chemotherapy(2017)
○Clinical Pharmacokinetics(2016)
○Seminar in Monitoring the Adverse Events of Cancer Chemotherapy(2016)
○Risk Management Practice for Cancer Patients(2016)



