Professor KATAOKA Kunishige
Faculty, Affiliation
Faculty of Chemistry, Institute of Science and Engineering
College and School Educational Field
Division of Material Chemistry, Graduate School of Natural Science and Technology
Division of Material Chemistry, Graduate School of Natural Science and Technology
Course in Chemistry, School of Chemistry, College of Science and Engineering
Laboratory
Biological Chemistry FAX:076-264-5742
Academic Background
【Academic background(Doctoral/Master's Degree)】
Kyoto University Doctor 199303 Unfinished course
【Academic background(Bachelor's Degree)】
University of Tsukuba 198803
Career
Osaka University Inst. for Scientific and Industrial Res.(1993/04/01-1994/12/31)
Osaka University Faculty of Science(1995/01/01-1996/03/31)
Osaka University Graduate School of Science(1996/04/01-2001/01/31)
Kanazawa University Faculty of Science(2001/02/01-2005/03/31)
Kanazawa University Grad. Sch. Natl Sci. & Tech.(2005/04/01-2011/03/31)
Kanazawa University Grad. Sch. Natl Sci. & Tech.(2011/04/01-)
Year & Month of Birth
1965/02
Academic Society
The Japanese Biochemical Society
The Japanese Biochemical Society
Japan Society for Bioscience, Biotechnology, and Agrochemistry
The Society of Biological Inorganic Chemistry
Award
○JB Award(1995/07/01)
○JB Award(1995/07/01)
○JB Award(1995/07/01)
○Nagase Science and Technology Foundation(2006/04/21)
Specialities
Functional biochemistry、Applied biochemistry、Bio-related chemistry、Biochemistry、Molecular Biology
Speciality Keywords
Biochemistry, Molecular Biology, Protein engineering, Metalloproteins
Research Themes
Books
- K. Kataoka, S. Yamashita etc. Copper in Biology: Molecular Structures, Cellular Processes and Living Systems (Edited by Yoshiaki Furukawa and Nobutaka Fujieda) Royal Society of Chemistry 2025/08
- T. Sakurai, K. Kataoka Copper-Oxygen Chemistry (Eds by K.D. Karlin and S. Itoh) John Wiley & Sons 2011/08
Papers
- Direct electron transfer-type bioelectrocatalytic dioxygen reduction with copper efflux oxidase lacking type I copper Adachi Taiki, Takei Toshitada, Kano Kenji, Yamashita Satoshi, Kataoka Kunishige, Sowa Keisei Electrochemistry Communications 179 108036 2025
- Bilirubin oxidase expression and activity enhancement from Myrothecium verrucaria in Aspergillus species Horiguchi Haruka Kado, Semba Hironori, Yamada Hiroyuki, Tsuboi Hirokazu, Bogaki Takayuki, Koda Akio, Kataoka Kunishige, Takagi Masahiro, Tsujino Yoshio Journal of Bioscience and Bioengineering 138 3 212-217 2024/09
- Structural insights into catalytic promiscuity of chalcone synthase from Glycine max (L.) Merr.: Coenzyme A-induced alteration of product specificity Waki Toshiyuki, Imaizumi Riki, Uno Kaichi, Doi Yamato, Tsunashima Misato, Yamada Sayumi, Mameda Ryo, Nakata Shun, Yanai Taro, Takeshita Kohei, Sakai Naoki, Kataoka Kunishige, Yamamoto Masaki, Takahashi Seiji, Nakayama Toru, Yamashita Satoshi Biochemical and Biophysical Research Communications 718 150080 2024/07/23
- Structural-Functional Correlations between Unique N-terminal Region and C-terminal Conserved Motif in Short-chain cis-Prenyltransferase from Tomato Imaizumi Riki, Matsuura Hiroaki, Yanai Taro, Takeshita Kohei, Misawa Shuto, Yamaguchi Haruhiko, Sakai Naoki, Miyagi-Inoue Yukino, Suenaga-Hiromori Miki, Waki Toshiyuki, Kataoka Kunishige, Nakayama Toru, Yamamoto Masaki, Takahashi Seiji, Yamashita Satoshi ChemBioChem 25 7 e202300796 2024/04/02
- Structural insights into a bacterial β-glucosidase capable of degrading sesaminol triglucoside to produce sesaminol: toward the understanding of the aglycone recognition mechanism by the C-terminal lid domain Yanai Taro, Takahashi Yukino, Katsumura Eri, Sakai Naoki, Takeshita Kohei, Imaizumi Riki, Matsuura Hiroaki, Hongo Shuntaro, Waki Toshiyuki, Takahashi Seiji, Yamamoto Masaki, Kataoka Kunishige, Nakayama Toru, Yamashita Satoshi Journal of Biochemistry 174 4 335-344 2023/10/01
- Influence of distal glycan mimics on direct electron transfer performance for bilirubin oxidase bioelectrocatalysts Nishida Sayaka, Sumi Hinata, Noji Haruna, Itoh Akira, Kataoka Kunishige, Yamashita Satoshi, Kano Kenji, Sowa Keisei, Kitazumi Yuki, Shirai Osamu Bioelectrochemistry 152 108413 2023/08
- Kinetic and thermodynamic analysis of Cu2+-dependent reductive inactivation in direct electron transfer-type bioelectrocatalysis by copper efflux oxidase Adachi Taiki, Mazurenko Ievgen, Mano Nicolas, Kitazumi Yuki, Kataoka Kunishige, Kano Kenji, Sowa Keisei, Lojou Elisabeth Electrochimica Acta 429 140987 2022/10/10
- Kinetic and thermodynamic analysis of Cu2+-dependent reductive inactivation in direct electron transfer-type bioelectrocatalysis by copper efflux oxidase Taiki Adachi, Ievgen Mazurenko, Nicolas Mano, Yuki Kitazumi, Kunishige Kataoka, Kenji Kano, Keisei Sowa, Elisabeth Lojou Electrochimica Acta 429 10 October 2022 140987 2022/08/09
- Effects of N-linked glycans of bilirubin oxidase on direct electron transfer-type bioelectrocatalysis Suzuki Yohei, Itoh Akira, Kataoka Kunishige, Yamashita Satoshi, Kano Kenji, Sowa Keisei, Kitazumi Yuki, Shirai Osamu Bioelectrochemistry 146 August 2022 108141 2022/04/25
- Structure-based engineering of a short-chain cis-prenyltransferase to biosynthesize nonnatural all-cis-polyisoprenoids: molecular mechanisms for primer substrate recognition and ultimate product chain-length determination Kutsukawa Ryo, Imaizumi Riki, Suenaga-Hiromori Miki, Takeshita Kohei, Sakai Naoki, Misawa Shuto, Yamamoto Masaki, Yamaguchi Haruhiko, Miyagi-Inoue Yukino, Waki Toshiyuki, Kataoka Kunishige, Nakayama Toru, Yamashita Satoshi, Takahashi Seiji FEBS Journal 2022/02/07
- Crystal structure of chalcone synthase, a key enzyme for isoflavonoid biosynthesis in soybean Riki Imaizumi, Ryo Mameda, Kohei Takeshita, Hiroki Kubo, Naoki Sakai, Shun Nakata, Seiji Takahashi, Kunishige Kataoka, Masaki Yamamoto, Toru Nakayama, Satoshi Yamashita, Toshiyuki Waki Proteins 89 1 126-131 2021/01/01
- Effects of elimination of α helix regions on direct electron transfer-type bioelectrocatalytic properties of copper efflux oxidase Taiki ADACHI, Yuki KITAZUMI, Osamu SHIRAI, Tenta KAWANO, Kunishige KATAOKA, Kenji KANO Electrochemistry 88 3 185-189 2020
- Diffusion-limited biosensing of dissolved oxygen by direct electron transfer-type bioelectrocatalysis of multi-copper oxidases immobilized on porous gold microelectrodes Miyata, M., Kitazumi, Y., Shirai, O., Kataoka, K., Kano, K. Journal of Electroanalytical Chemistry 860 2020
- Roles of the indole ring of Trp396 covalently bound with the imidazole ring of His398 coordinated to type I copper in bilirubin oxidase Kataoka, K., Ito, T., Okuda, Y., Sakai, Y., Yamashita, S., Sakurai, T. Biochemical and Biophysical Research Communications 521 3 620-624 2020
- A bio-solar cell with thylakoid membranes and bilirubin oxidase Adachi, T., Kataoka, K., Kitazumi, Y., Shirai, O., Kano, K. Chemistry Letters 48 7 686-689 2019
- Structural changes of the trinuclear copper center in bilirubin oxidase upon reduction Tokiwa, T., Shoji, M., Sladek, V., Shibata, N., Higuchi, Y., Kataoka, K., Sakurai, T., Shigeta, Y., Misaizu, F. Molecules 24 1 2018/12/26
- Redox Potential-Dependent Formation of an Unusual His–Trp Bond in Bilirubin Oxidase Akter, M., Tokiwa, T., Shoji, M., Nishikawa, K., Shigeta, Y., Sakurai, T., Higuchi, Y., Kataoka, K., Shibata, N. Chemistry - A European Journal 24 68 18052-18058 2018
- Amino acids located in the outer-sphere of the trinuclear copper center in a multicopper oxidase, CueO as the putative electron donor in the four-electron reduction of dioxygen Sakurai, T, Yamamoto, M., Ikeno, S., Kataoka, K. Biochimica et Biophysica Acta - Proteins and Proteomics 1865 8 997-1003 2017
- Heterologous expression of Halomonas halodenitrificans nitric oxide reductase and its N-terminally truncated NorC subunit in Escherichia coli Sakurai, N., Kataoka, K., Sugaya, N., Shimodaira, T., Iwamoto, M., Shoda, M., Horiuchi, H., Kiyono, M., Ohta, Y., Triwiyono, B., Seo, D., Sakurai, T. Journal of Inorganic Biochemistry 169 1 61-67 2017
- Biochemical, spectroscopic and X-ray structural analysis of deuterated multicopper oxidase CueO prepared from a new expression construct for neutron crystallography Akter, M., Inoue, C., Komori, H., Matsuda, N., Sakurai, T., Kataoka, K., Higuchi, Y., Shibata, N. Acta Crystallographica Section:F Structural Biology Communications 72 788-794 2016
- Exogenous acetate ion reaches the type II copper centre in CueO through the water-excretion channel and potentially affects the enzymatic activity Komori, H., Kataoka, K., Tanaka, S., Matsuda, N., Higuchi, Y., Sakurai, T. Acta Crystallographica Section:F Structural Biology Communications 72 558-563 2016
- A pirin-like protein from Pseudomonas stutzeri and its quercetinase activity Widiatningrum, T., Maeda, S., Kataoka, K., Sakurai, T. Biochemistry and Biophysics Reports 3 144-149 2015
- A novel resting form of the trinuclear copper center in the double mutant of a multicopper oxidase, CueO, Cys500Ser/Glu506Ala Takao Kajikawa, Ryosuke Sugiyama, Kunishige Kataoka, Takeshi Sakurai Journal of Inorganic Biochemistry 149 88-90 2015
- New Insights into the Catalytic Active-Site Structure of Multicopper Oxidases H. Komori, R. Sugiyama, K. Kataoka, K. Miyazaki, Y. Higuchi and T. Sakurai Acta Crystallographica Section D: Biological Crystallography D70 772-729 2014
- Spectroscopic and Electrochemical Properties of Cytochrome c551 from Alcaligenes xylosoxidans GIFU 1051. Bulletin of Chemical Society of Japan 73 8 1839-1840 2000
- Functional Analysis of Conserved Aspartate and Histidine Residues Located Around the Type 2 Copper Site of Copper-Containing Nitrite Reductase. JOURNAL OF BIOCHEMISTRY 127 2 345-350 2000
- Crystallization and Preliminary X-ray Crystallographic Studies of Mavicyanin from Cucurbita pepo medullosa. ACTA CRYSTALLOGRAPHICA SECTION D-BIOLOGICAL CRYSTALLOGRAPHY 59 1474-1476 2003
- Crystallization and Preliminary X-ray Crystallographic Studies of Dissimilatory Nitrite Reductase Isolated from Hyphomicrobium denitrificans A3151. ACTA CRYSTALLOGRAPHICA SECTION D-BIOLOGICAL CRYSTALLOGRAPHY 60 2383-2386 2004
- Modifications on the Hydrogen Bond Network by Mutations of Escherichia coli Copper Efflux Oxidase Affect the Process of Proton Transfer to Dioxygen Leading to Alterations of Enzymatic Activities T. Kajikawa, K. Kataoka, and T. Sakurai Biochem. Biophys. Res. Commun. 422 152-156 2012
- Study on dioxygen reduction by mutational modifications of the hydrogen bond network leading from bulk water to the trinuclear copper center in bilirubin oxidase Morishita, Hirotoshi; Kurita, Daisuke; Kataoka, Kunishige; Sakurai, Takeshi Biochemical and Biophysical Research Communications 450 1 767-772 2014

- Electrochemical Characterization of a Unique "Neutral" Laccase from Flammulina velutipes K. Saito, S. Kurose, Y. Tsujino, T. Osakai, K. Kataoka, T. Sakurai, and E. Tamiya J. Biosci. Bioeng. 115 159-167 2013
- Modifications of Laccase Activities of Copper Efflux Oxidase, CueO by Synergistic Mutations in the First and Second Coordination Spheres of the Type I Copper Center K. Kataoka, H. Kogi, S. Tsujimura, and T. Sakurai Biochem. Biophys. Res. Commun. 431 393-397 2013
- Role of the Hydrogen Bond Connecting the Ligands for Substrate and Type I Copper in the Cuprous Oxidase, CueO K. Kataoka and T. Sakurai Chem. Lett. 42 1102-1104 2013
- Crystal Structure of the CueO Mutants at Glu506, the Key Amino Acid Located in the Proton Transfer Pathway for Dioxygen Reduction H. Komori, T. Kajikawa, K. Kataoka, Y. Higuchi, and T. Sakurai Biochem. Biophys. Res. Commun. 438 686-690 2013
- An O-Centered Structure of the Trinuclear Copper Center in the Cys500Ser/Glu506Gln Mutant of CueO and Structural Changes in Low to High X-Ray Dose Conditions. H. Komori, R. Sugiyama, K. Kataoka, Y. Higuchi, and T. Sakurai ANGEWANDTE CHEMIE-INTERNATIONAL EDITION 51 1861-1864 2012/01
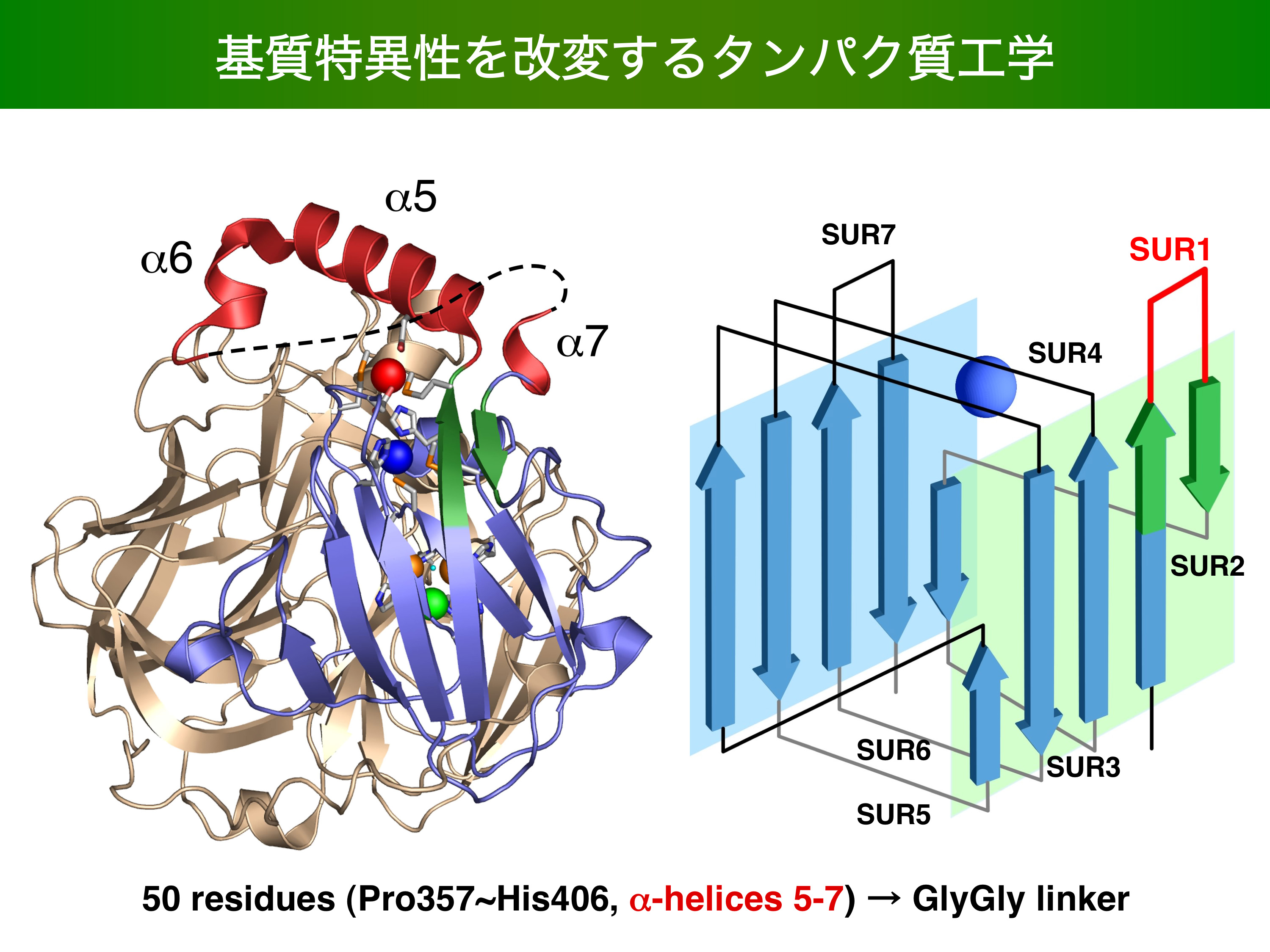
- Enhancement of Laccase Activity through the Construction and Breakdown of a Hydrogen Bond at the Type I Copper Center in Escherichia coli CueO and the Deletion Mutant Δα5-7 CueO. K. Kataoka, T. sakura et al. BIOCHEMISTRY 50 558-565 2011/08
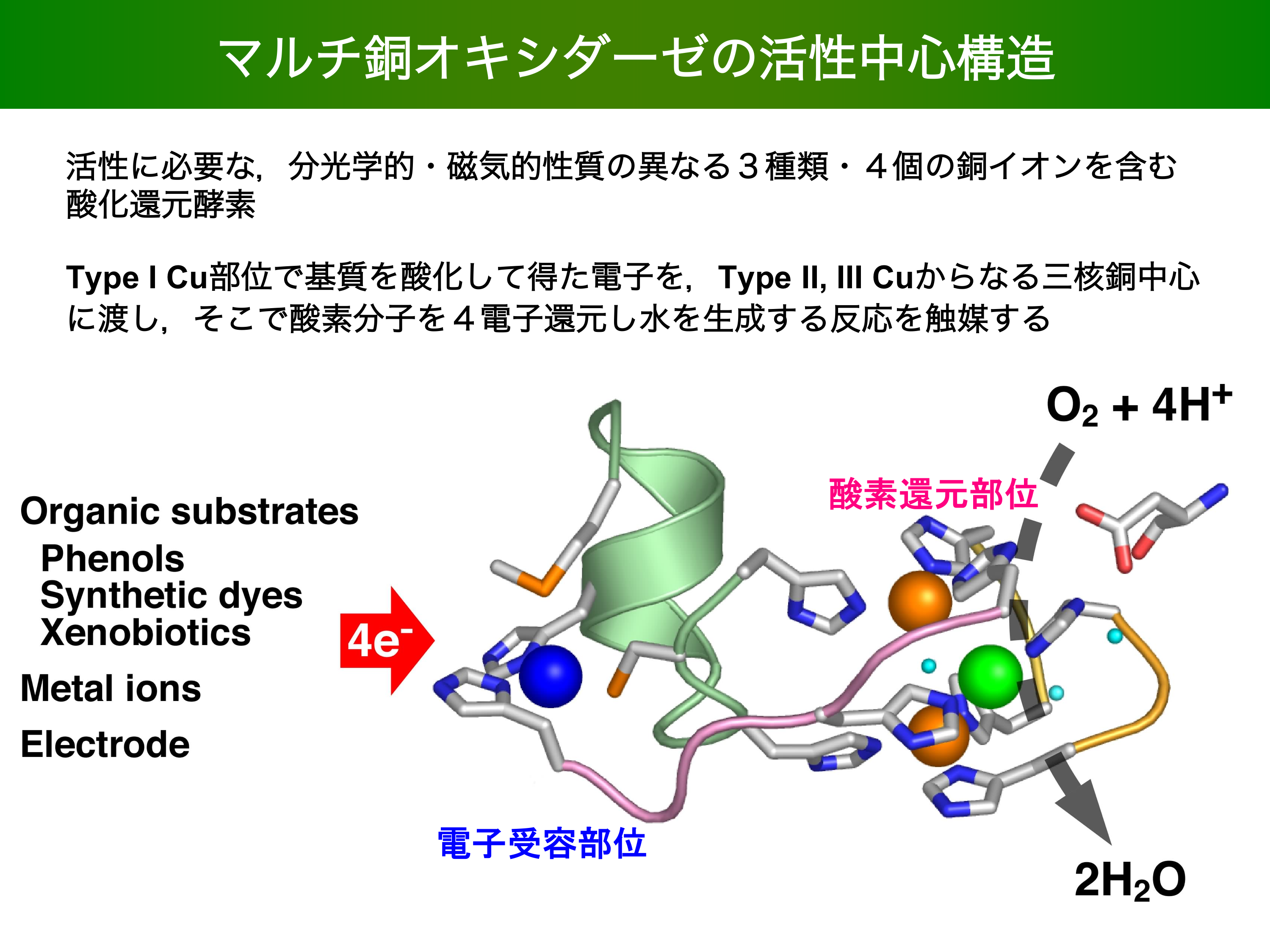
- Four-Electron Reduction of Dioxygen by a Multicopper Oxidase, CueO, and Roles of Asp112 and Glu506 Located Adjacent to the Trinuclear Copper Center K. Kataoka, T. Sakurai et al. JOURNAL OF BIOLOGICAL CHEMISTRY 284 14405-14413 2009/05
- Compensatory Binding of an Asparagine Residue to the Coordination-Unsaturated Type I Cu Center in Bilirubin Oxidase Mutants. K. Kataoka, T. Sakurai et al. BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS 371 416-419 2008/04
- ATR-FTIR Study of the Protonation States of Glu Residue in the Multicopper Oxidase, CueO and Bilirubin Oxidase. M. Iwaki, K. Kataoka, T. Kajino, R. Sugiyama, H. Morishita, and T. Sakurai FEBS LETTERS 584 4027-4031 2010/08
- Mutation at Asp112 Adjacent to the Trinuclear Cu Center in CueO as the Proton Donor in the Four-Electron Reduction of Dioxygen Y. Ueki, K. Kataoka et al. FEBS LETTERS 580 4069-4072 2006/06
- Tandem and Single Genes of Three Membrane-Bound Nitrate Transporters in the nar Gene Cluster of the Moderately Halophilic Denitrifier, Halomonas halodenitrificans. DNA SEQUENCE 17 363-369 2006/10
- Structure and Function of a Hexameric Copper-Containing Nitrite Reductase. N. Nojiri, Y. Xie, K. Kataoka, S. Suzuki et al. PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AME 104 4315-4320 2007/03
- Effect of Axial Ligand Mutation of the Type I Copper Site in Bilirubin Oxidase on Direct Electron Transfer-Type Bioelectrocatalytic Reduction of Dioxigen. Electoanalytical chemistry 601 119-124 2007/03
- Probing Electron Transfer Reactions between Two Azurins from Alcaligenes xylosoxidans GIFU 1051 with Optically Active Ru Complexes as Molecular Recognition Probes: Importance of the 43rd Residue. INORGANICA CHIMICA ACTA 360 1555-1567 2007/08
- Bioelectrocatalytic Reduction of O2 Catalyzed by CueO from Escherichia coli Adsorbed on a Highly Oriented Pyrolytic Graphite Electrode. CHEMISTRY LETTERS 36 132-133 2007/01
- Promotion of Laccase Activities of Escherichia coli Cuprous Oxidase, CueO by Deleting the Segment Covering the Substrate Binding Site. CHEMISTRY LETTERS 36 232-233 2007/02
- A Novel Zinc-Dependent D-Serine Dehydratase from Saccharomyces cerevisiae T. Ito, K. Kataoka, T. Yoshimura et al. BIOCHEMICAL JOURNAL 409 399-406 2008/01
- Measurement of Electric Field Gradient at 117In on the Cu-site in Mavicyanin by Perturbed Angular Correlation of &gamma-rays A. Yokoyama, K. Kataoka, Y. Ohkubo et al. HYPERFINE INTERACTIONS 181 69-73 2008/08
- Direct Electrochemistry of CueO and Its Mutants at Residues to and near Type I Cu for Oxygen-Reducing Biocathode. Y. Miura, S. Tsujimura, K. Kataoka et al. Fuel Cells 9 70-78 2009/01
- Studies of Interaction of Homo-Dimeric Ferredoxin-NAD(P)+ Oxidoreductases of Bacillus subtiis and Rhodopseudomonas palustris, That Are Closely Related to Thioredoxin Reductase in Amino Acid Sequence, with Ferredoxins and Pyridine Nucleotide Coenzymes. D. Seo, S. Okabe, K. Kataoka et al. BIOCHIMICA ET BIOPHYSICA ACTA-PROTEIN STRUCTURE AND MOLECULAR ENZYMOLOGY 1794 594-601 2009/01
- Modification of Spectroscopic Properties and Catalytic Activity of Escherichia coli CueO by Mutations of Methionine 510, the Axial Ligand to the Type I Cu S. Kurose, K. Kataoka et al. BULLETIN OF THE CHEMICAL SOCIETY OF JAPAN 82 504-508 2009/04
- Characterization of Two Cu-Containing Protein Fragments Obtained by Limited Ptoteolysis of Hyphomicrobium denitrificans A3151 Nitrite Reductase. BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS 300 1 36-40 2003/01
- Point Mutations at the Type I Cu Ligands, Cys457 and Met467, and at the Putative Proton Donor, Asp105, in Myrothecium verrucaria Bilirubin Oxidase and Reactions with Dioxygen. K. Kataoka, T. sakura et al. BIOCHEMISTRY 44 18 7004-7012 2005
- Structure and Function of the Engineered Multicopper Oxidase, CueO from Escherichia coli - Deletion of the Methionine-Rich Helical Region Covering the Substrate Binding Site -. K. Kataoka, H. Komori, Y. Higuchi, T. Sakurai et al. JOURNAL OF MOLECULAR BIOLOGY 373 141-152 2007
- Authentic and Recombinant Bilirubin Oxidase Are in Different Resting Forms. BIOSCIENCE BIOTECHNOLOGY AND BIOCHEMISTRY 67 5 1157-1159 2003/05
- Structure of Azurin I from the Denitrifying Bacterium Alcaligenes xylosoxidans NCIMB 11015 at 2.45 A Resolution. Acta Crystallographica 54 347-354 1998
- Crystal Structure Determination of Oxidized and Reduced Pseudoazurin from Achromobacter cycloclastes. The Journal of Biological Chemistry 274 25 17845-17852 1999/06
- High-Level Expression of Myrothecium verrucaria Bilirubin Oxidase in Pichia pastoris, and its Facile Purification and Characterization. K. Kataoka, T. sakurai et al. PROTEIN EXPRESSION AND PURIFICATION 41 77-83 2005
- The Reversible Change in the Redox State of Type I Cu in Myrothecium verrucaria Bilirubin Oxidase Depending on pH. BIOSCIENCE BIOTECHNOLOGY AND BIOCHEMISTRY 69 9 1998-2000 2004
- Characterization and Function of Met150Gln Mutant of Copper-Containing Nitrite Reductase from Achromobacter cycloclastes. BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS 303 519-524 2003/03
- Primary Structure of a Japanese Lacquer Tree Laccase as a Prototype Enzyme of Multicopper Oxidases. JOURNAL OF INORGANIC BIOCHEMISTRY 91 1 125-131 2002/07
- Matrix-assisted Laser Desorption/Ionization and Electrospray Ionization Mass Spectrometry Analysis of Blue Copper Proteins. Azurin and Mavicyanin RAPID COMMUNICATIONS IN MASS SPECTROMETRY 12 1967-1971 1998
- Spectroscopic and Electrochemical Properties of the Met86Gln Mutant of Achromobacter cycloclastes Pseudoazurin. JOURNAL OF INORGANIC BIOCHEMISTRY 82 79-84 2000
- The Significance of the Flexible Loop in the Azurin (Az-iso2) from the Obligate Methylotroph Methylomonas sp. Strain J. JOURNAL OF MOLECULAR BIOLOGY 333 117-124 2003
- Characterization of Two Type 1 Cu Sites of Hyphomicrobium denitrificans Nitrite Reductase: A New Class of Copper-containing Nitrite Reductases. BIOCHEMISTRY 43 44 14180-14188 2004
- Spectroscopic and Electrochemical Properties of Two Azurins (Az-iso1 and Az-iso2) from the Obligate Methylotroph Methylomonas sp. Strain J and the Structure of Novel Az-iso2. JOURNAL OF BIOLOGICAL INORGANIC CHEMISTRY 4 749-758 1999
- Structure-Based Engineering of Alcaligenes xylosoxidans Copper-Containing Nitrite Reductase Enhances Intermolecular Electron Transfer Reaction with Pseudoazurin. JOURNAL OF BIOLOGICAL CHEMISTRY 279 51 53374-53378 2004
- Crystallization and Preliminary X-ray Study of iso-2 Azurin from the Methyloyrophic Bacterium, Methylomonas J. Acta Crystallographica 55 307-309 1999
- Intramolecular Electron Transfer Process of Native and Mutant Forms of Blue Copper-Containing Nitrite Reductase from Alcaligenes xylosoxidans. Inorganic Reaction Mechanisms 2 129-135 2000
- Diverse NO Reduction by Halomonas halodenitrificans Nitric Oxide Reductase. T. Sakurai, K. Kataoka et al. BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS 333 483-487 2005
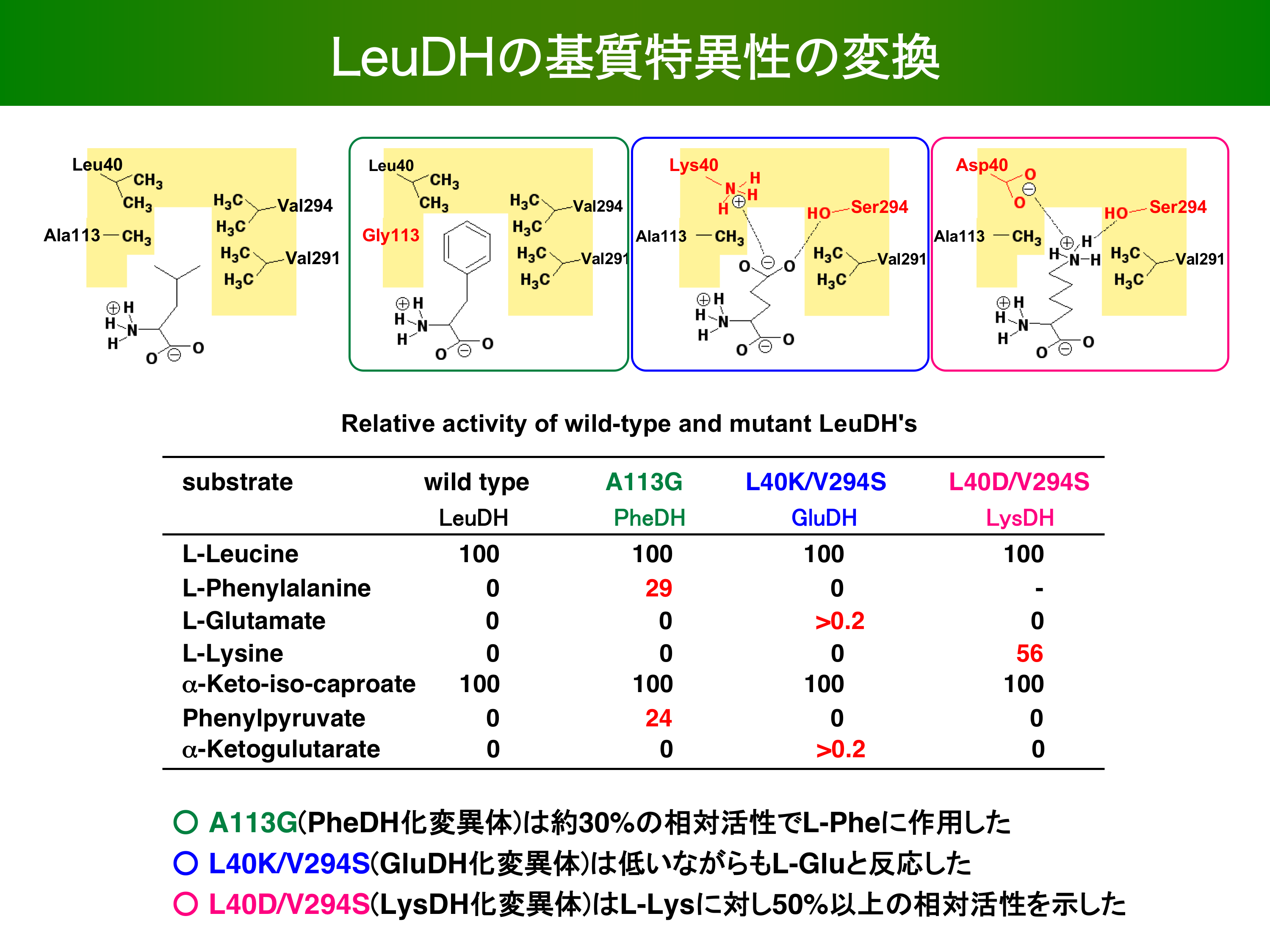
- Alteration of Substrate Specificity of Leucine Dehydrogenase by Site-Directed Mutagenesis. JOURNAL OF MOLECULAR CATALYSIS B-ENZYMATIC 23 299-309 2003
- Structural Reorganization of Copper Binding Site Involving Thr15 of Mavicyanin from Cucurbita pepo medullosa (Zucchini) upon Reduction. Y. Xie, T. Inoue, K. Kataoka et al. JOURNAL OF BIOCHEMISTRY 137 455-461 2005
- Spectroscopic and Functional Characterization Cu-Containing Nitrite Reductase from Hyphomicrobium denitrificans A3151. JOURNAL OF INORGANIC BIOCHEMISTRY 91 1 132-138 2002/07
- Characterization of Azide-binding Type 2 Cu(II) Site of Nitrite Reductase. Bulletin of Chemical Society of Japan 74 1 183-184 2001/01
- Fragmentary Form of Thermostable Leucine Dehydrogenase of Bacillus stearothermophilus : Its Construction and Reconstitution of Active Fragmentary Enzyme. Biochemical Biophysical Research Communications 280 4 1177-1182 2001
- Spectroscopic Distinction between Two Co(II) ions Substituted for Type 1 and 2 Cu in Nitrite Reductase. Inorganica Chemica Acta 275 289-294 1998
- Type 1 Cu Structure of Blue Nitrite Reductase from Alcaligenes xylosoxidans GIFU 1051 at 2.05 A Resolution: Comparison of Blue and Green Nitrite Reductase. JOURNAL OF BIOCHEMISTRY 124 5 876-879 1998/05
- Gene Synthesis, Expression, and Mutagenesis of Zucchini Mavicyanin: The Fourth Ligand of Blue Copper Center Is Gln. Biochemical Biophysical Research Communications 250 2 409-413 1998
- Crystallization and Preliminary X-ray Studies of Azurin-I and Azurin-II from Denitrifying Bacterium Alcaligenes xylosoxidans GIFU1051. Acta Crystallographica 53 337-338 1997
- Spectroscopic Characterization and Intramolecular Electron Transfer Processes of Native and Type 2 Cu-Depleted Nitrite Reductases. JOURNAL OF BIOLOGICAL INORGANIC CHEMISTRY 2 265-274 1997
Conference Presentations
- Heterologous expression and purification of the multicopper oxidase from Clostridium beijerinckii(2019/09/19)
- Enhancement of catalytic activity of a cuprous oxidase, CueO by random and site-directed mutagenesis (2019/03/26)
- Roles of the Trp396-His398 cross-linkage formed in the type I copper center in bilirubin oxidase(2017/03/17)
- Enhancement of laccase activities of the copper efflux oxidase, CueO by tuning the redox potential of the type I copper center(conference:The 5th International Conference on Cofactors and Active Enzyme Molecule 2016)(2016/09/04)
- The activity control of the multi-copper oxidase by the precise control of the redox potential of the type I copper site(2016/03/27)
- Structural studies to elucidate substrate recognition mechanisms of sesaminol glycoside hydrolases(2025/03/07)
- Investigating the mechanism of higher-order structure formation and X-ray crystallography of chalcone isomerase from snapdragon(2025/03/07)
- Coordination structure of the typeIcopper site of laccase from Clostridium beijerinckii(2025/03/07)
- Activation of bilirubin oxidase expressed in Escherichia coli by ascorbic acid treatment under anaerobic conditions(2025/03/07)
Others
- Metal Coordination and Mechanism of Multicopper Nitrite Reductase. 33 10 728-735 2000
- Structure-Function Relationships of Copper-Containing Nitrite Reductase. 190 245-265 1999
- Stracture and Function of Multicopper Oxidoreductases. 77 2 148-153 2005/02
- Structure and Function of CueO, a Multicopper Oxidase Concerned in Homeostasis of Copper. 17 3 308-315 2006
- Basic and Applied Features of Multicopper Oxidases, CueO, Bilirubin Oxidase, and Laccase. 7 220-229 2007
- Structure and Function of Type I Copper in Multicopper Oxidases. T. Sakurai, K. Kataoka Birkhauser 64 2642-2656 2007
Arts and Fieldwork
Patent
Theme to the desired joint research
○Industrial application of multi-copper oxidase
○Elucidation and application of the microbial denitrification mechanism.
Grant-in-Aid for Scientific Research
○「古代色素「貝紫」生合成酵素の高選択的臭素化反応メカニズムの解明と応用」(2015-2017)
○「バイオ燃料電池の実用化に必須である高反応性カソード電極用酵素の研究開発」(2011-2013)
○「金属タンパク質をホロ化する金属シャペロンタンパク質の開発」(2009-2010)
○「金属タンパク質中の活性位における超微細電場測定」(2004-2005)
○「一酸化窒素還元から酸素還元への変換による呼吸酵素の分子進化」(2004-2005)
○「一酸化窒素還元酵素の活性中心構造と触媒反応機構の解明」(2002-2003)
○「銅型亜硝酸還元酵素の構造と触媒反応機構および電子伝達機能に関する研究」(1999-2000)
○「マルチヘム型一酸化窒素還元酵素を用いたプロトンポンプ分子進化過程の解明」(2007-2009)
○「タンパク質工学によるアミノ酸脱水素酵素の基質特異性の変換」(1995-1995)
Competitive research funding,Contribution
Collaborative research,Consignment study
○Strategic Development of PEFC Technologies for Practical Application. Development of Technology for Next-generation Fuel Cells. Research and Development of Novel Anode Catalysts Based on Multi-copper Oxidases(2005-2009)
Classes (Bachelors)
○Experiments in Basic Chemistry A(2020)
○Biochemistry III(2020)
○Experiments in Basic Chemistry C(2020)
○Biochemistry I(2020)
○Safety in Chemistry(2020)
○Introduction of Chemistry A(2020)
○Introduction of Chemistry B(2020)
○Practical Seminar for Teaching Profession C / Junior High School・High School(2020)
○Introduction of Chemistry A(2019)
○Safety in Chemistry(2019)
○Biochemistry I(2019)
○Biochemistry III(2019)
○Experiments in Basic Chemistry C(2019)
○Teaching Practicum IV(2019)
○Specialized Topics in Chemistry(2018)
○Experiments in Basic Chemistry C(2018)
○Biochemistry III(2018)
○Practical Seminar for Teaching Profession C / Junior High School・High School(2018)
○Biochemistry I(2018)
○Safety in Chemistry(2018)
○Experiments in Basic Chemistry C(2017)
○Biochemistry III(2017)
○Introduction of Chemistry A(2017)
○Practical Seminar for Teaching Profession C / Junior High School・High School(2017)
○Biochemistry I(2017)
○Introduction of Chemistry B(2017)
○Safety in Chemistry(2017)
○Safety in Chemistry(2016)
○Biochemistry I(2016)
○Biochemistry III(2016)
○Experiments in Basic Chemistry C(2016)
○Practical Seminar for Teaching Profession C / Junior High School・High School(2016)
Classes (Graduate Schools)
○Biochemistry for Education(2020)
○Seminar in Material Creation(2020)
○Molecular Enzyme Chemistry(2020)
○Material Creation Chemistry IV(2020)
○Seminar in Material Creation(2020)
○Material Creation Chemistry IV(2019)
○Seminar in Material Creation(2019)
○Molecular Enzyme Chemistry(2019)
○Seminar in Material Creation(2019)
○Biochemistry for Education(2019)
○Molecular Enzyme Chemistry(2018)
○Protein Engineering(2018)
○Seminar in Material Creation(2018)
○Topics in Chemistry(2018)
○Material Creation Chemistry IV(2018)
○Protein Engineering(2017)
○Protein Engineering(2017)
○Protein Engineering(2017)
○Seminar in Material Creation(2017)
○Material Creation Chemistry IV(2017)
○Seminar in Material Creation(2017)
○Molecular Enzyme Chemistry(2017)
○Protein Engineering(2017)
○Seminar in Material Creation(2016)
○Material Creation Chemistry IV(2016)
○Molecular Enzyme Chemistry(2016)
○Seminar in Material Creation(2016)
○Protein Engineering(2016)